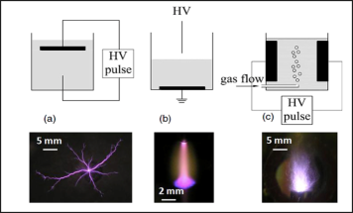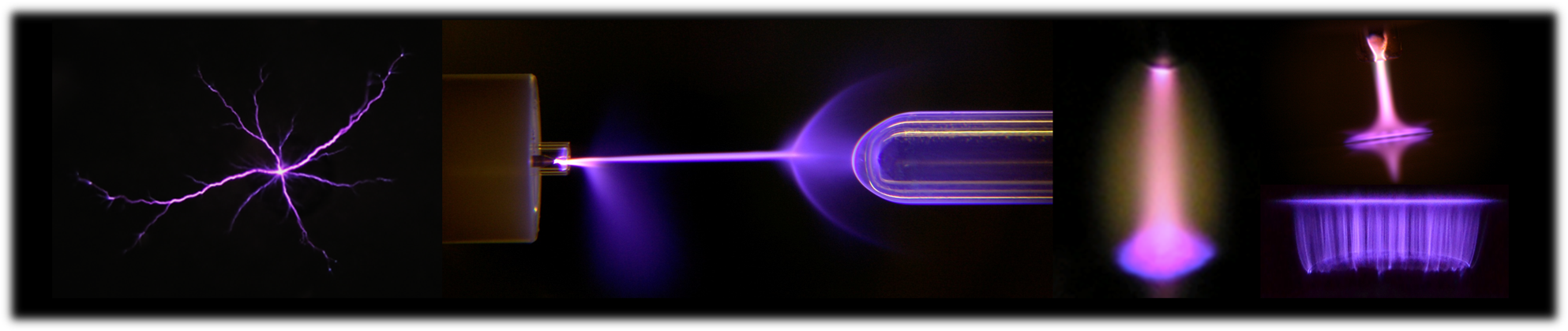
Interactions between different types of plasma and liquid
Non-equilibrium plasmas formed either directly in liquid water or in a gas phase interacting with a liquid phase is of growing interest for many applications. The process of converting electrical energy into useful chemical species by plasmas is of specific interest as radicals and molecules formed from water and solutes initiate a wide variety of useful biochemical and chemical reactions.
Increasing research efforts have recently been devoted to the plasma-liquid interaction, but there remain many unresolved questions about the properties of plasma interaction with liquids. Two key challenges in this field are the breakdown processes and mechanisms in liquids and the physical and chemical processes occurring at the plasma-liquid interface. The second challenge is particularly multi-disciplinary due to the wide range of chemical species and physical effects which involve radical and reactive species, ions, electrons (V)UV emission, electric fields, heat, and neutral gas flows across the gas-liquid interface. All these individual components are typically studied in distinct fields of research.
In our group we study (a) direct discharges in liquids, (b) gas phase discharges interacting with a liquid such as DC glow discharges and plasma jets, and (c) discharges generated inside bubbles. Applications of interest include water treatment, disinfection, and nanoparticle synthesis.
Further reading:
- P. Bruggeman and C. Leys. “Non-thermal plasmas in and in contact with liquids,” Invited topical review paper, J. Phys. D: Appl. Phys., 42 (2009), 053001.
- Y. Yue, V.S.S.K. Kondeti, and P.J. Bruggeman. “Absolute atomic hydrogen density measurements in an atmospheric pressure plasma jet: generation, transport and recombination from the active discharge region to the effluent,” Plasma Sources Sci. Technol., 29(4) (2020), 04LT01.